1. What is an AID system?
An automated insulin delivery (AID) system consists of three main parts: 1) a continuous glucose monitor (CGM) sensor, 2) an insulin pump, and 3) an algorithm. These three parts “talk” to each other, so the pump can automatically give you the right amount of insulin based on your glucose levels.
2. How is AID different from other methods of insulin delivery?
For people who need both meal-time (bolus) and long-acting (basal) insulin, there are a few broad categories of methods for delivering insulin:
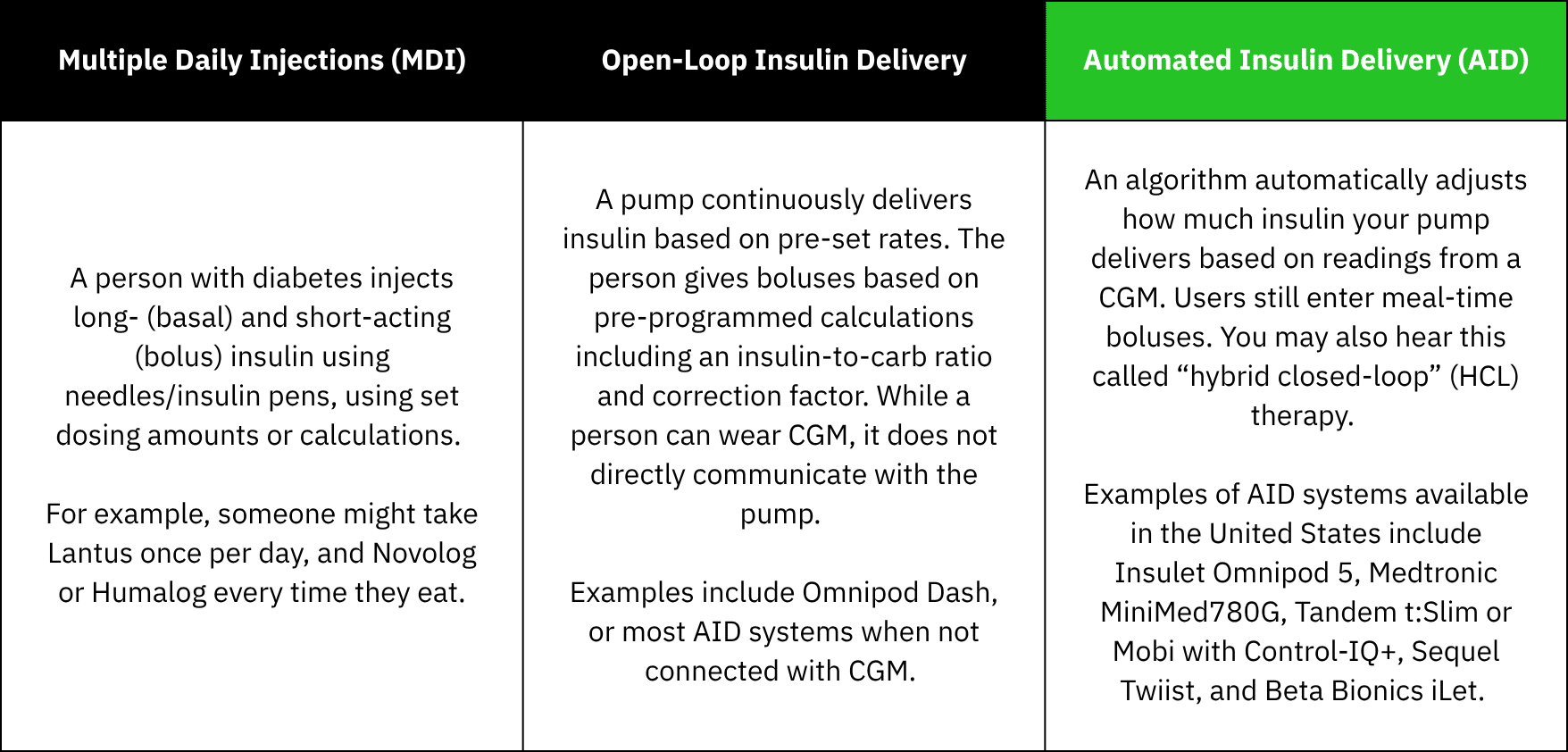
The biggest thing that sets AID apart is how it helps to adjust glucose levels without requiring as much action and attention from the user to stay in range.
3. Who is it for?
The American Diabetes Association recommends AID for people with type 1 diabetes, people with type 2 diabetes on basal + mealtime insulin treatment, and people with other forms of insulin-deficient diabetes (LADA and others). They also say AID should be considered for people on basal insulin only, who have barriers to reaching their targets.1
Outside of pregnancy, all AID systems are approved for use in type 1 diabetes. For type 2 diabetes, Omnipod 5, MiniMed 780G, and t:Slim x2 or Mobi pumps with the Control-IQ+ algorithm are approved.
Research on AID in pregnancy has focused on people with type 1 diabetes. Experts have called for much-needed research on AID use in pregnancy with type 2 diabetes and are waiting to make recommendations for this group.
The bottom line is this: no matter what kind of diabetes you have, you deserve to know what tools are available, so that you can discuss them with your healthcare team and speak up about what you think is best for your needs.
4. What’s so great about AID?
Outside of pregnancy, studies show that AID systems are the best available tools to meet blood sugar targets, helping people with type 1 diabetes spend 12% more time in range on average than with non-automated insulin pumps or multiple daily injections.2–4
In pregnancy with type 1 diabetes, some AID systems have been shown to increase time spent in the pregnancy target range (63–140 mg/dL) by up to 15% on average compared to MDI or open-loop pump therapy with a CGM.5,6 This is key, because every additional 5% time in pregnancy range has been shown to reduce risks for you and your baby.7
5. Is AID safe to use during pregnancy?
Studies have found that available AID systems can safely manage glucose during pregnancy with type 1 diabetes without increasing risk for dangerous lows or highs.5,8–13 As of early 2026, three AID systems—Medtronic MiniMed 780G, CamDiab CamAPS FX, and Tandem t:Slim X2 with Control-IQ—have been studied in large trials of pregnant people with type 1 diabetes, which give experts more confidence that these systems can safely be used in pregnancy.5,8,9 In the United States, the Tandem t:Slim X2 and Mobi systems with Control-IQ+ technology are the only available AID systems approved by the FDA specifically for use in pregnancy. CamAPS has been approved for use in pregnancy by many regulatory bodies globally (including FDA), but it is currently only available outside of the United States.
Importantly, some AID systems have been studied in pregnancy more than others, so we still have lots to learn. Research on many AID systems in type 1 diabetes is still ongoing, and none have been studied specifically for pregnancy with type 2 diabetes. Still, many people with diabetes and their care teams have used the available systems during pregnancy, and found them to be very helpful.
6. Which AID systems are recommended for pregnancy?
If possible, experts recommend using an AID system that meets the following three criteria:14
- Studied in a large randomized controlled trial (the standard for high-quality medical research) of pregnant people
- Shown in a randomized controlled trial to increase time in the target pregnancy range by at least 5% per day (compared to non-AID methods of insulin delivery)
- Has the ability to adapt to insulin sensitivity changes in pregnancy (either with a pregnancy-specific glucose target and/or algorithm features.)
Currently, CamAPS is the only AID system that fits all three criteria, but is not yet available in the United States. Other options which meet at least one criteria include Tandem t:Slim X2 and Mobi with Control-IQ+ (meets criteria 1 and 2), Medtronic MiniMed 780G (meets criteria 1), and Sequel twiist (meets criteria 3). The biggest differences between available systems to consider are how much you and your healthcare team will need to adjust settings and dosing strategies (beyond how you typically would outside of pregnancy), and whether the system has been shown to increase the time you spend in the target glucose range for pregnancy.
If you aren’t able to access a recommended system, don’t fret. There may be settings you can change or new dosing strategies you can try to help other systems meet the unique needs of pregnancy. It’s important to have support when making these changes—talk with your healthcare team to explore what’s best for you (and check out these expert tips together!)
7. Can I keep using my current AID system during pregnancy?
Yes! Ideally, experts recommend starting a system that fits the three criteria mentioned above before pregnancy, when planning or trying to conceive. This will help you feel as prepared as possible going into pregnancy, and give you the best support for your management without having to worry about switching and learning something new amidst all of the other changes pregnancy brings. But remember—if you become pregnant while using a different AID system or can’t access a new system, your healthcare team can help you optimize the settings on your current system to support your needs during pregnancy.
8. I don’t use AID, and I just found out I’m pregnant. Is it too late to switch?
No, it’s not! Experts recommend starting AID as soon as possible in pregnancy. Tell your healthcare team about your pregnancy, and ask about what AID systems are available to you (you can find more information on insurance coverage here). We wish you a happy, healthy pregnancy—with less diabetes on your mind, thanks to a little help from AID.
References
- American Diabetes Association Professional Practice Committee for Diabetes*. 7. Diabetes Technology: Standards of Care in Diabetes—2026. Diabetes Care. 2025;49(Supplement_1):S150-S165. doi:10.2337/dc26-S007
- Boughton CK, Hovorka R. New closed-loop insulin systems. Diabetologia. 2021;64(5):1007-1015. doi:10.1007/s00125-021-05391-w
- Leelarathna L, Choudhary P, Wilmot EG, et al. Hybrid closed-loop therapy: Where are we in 2021? Diabetes Obes Metab. 2021;23(3):655-660. doi:10.1111/dom.14273
- Phillip M, Nimri R, Bergenstal RM, et al. Consensus Recommendations for the Use of Automated Insulin Delivery Technologies in Clinical Practice. Endocr Rev. 2023;44(2):254-280. doi:10.1210/endrev/bnac022
- Donovan LE, Lemieux P, Dunlop AD, et al. Closed-Loop Insulin Delivery in Type 1 Diabetes in Pregnancy: The CIRCUIT Randomized Clinical Trial. JAMA. Published online October 24, 2025. doi:10.1001/jama.2025.19578
- Lee TTM, Collett C, Bergford S, et al. Automated Insulin Delivery in Women with Pregnancy Complicated by Type 1 Diabetes. N Engl J Med. 2023;389(17):1566-1578. doi:10.1056/NEJMoa2303911
- Murphy HR. Continuous glucose monitoring targets in type 1 diabetes pregnancy: every 5% time in range matters. Diabetologia. 2019;62(7):1123-1128. doi:10.1007/s00125-019-4904-3
- Lee TT, Collett C, Bergford S, et al. Automated closed-loop insulin delivery for the management of type 1 diabetes during pregnancy: the AiDAPT RCT. Published online 2024. Accessed September 8, 2024. http://www.ncbi.nlm.nih.gov/books/NBK603527/
- Benhalima K, Beunen K, Van Wilder N, et al. Comparing advanced hybrid closed loop therapy and standard insulin therapy in pregnant women with type 1 diabetes (CRISTAL): a parallel-group, open-label, randomised controlled trial. Lancet Diabetes Endocrinol. 2024;12(6):390-403. doi:10.1016/S2213-8587(24)00089-5
- Tahir S, Naeem S, Nayyab I, et al. Hybrid closed loop insulin therapy versus standard therapy in pregnant women with type 1 diabetes: A systematic review and meta-analysis of randomized controlled trials. Eur J Obstet Gynecol Reprod Biol. 2025;310:113969. doi:10.1016/j.ejogrb.2025.113969
- Stamati A, Christoforidis A. Automated insulin delivery in pregnant women with type 1 diabetes mellitus: a systematic review and meta-analysis. Acta Diabetol. 2025;62(4):441-452. doi:10.1007/s00592-025-02446-x
- Pham AQ, Lingvay I, Ahmadi N, Santiago-Munoz PC, Abreu M. Use of commercially available automated insulin delivery systems in pregnant people with type 1 diabetes. Diabetes Res Clin Pract. 2025;228:112454. doi:10.1016/j.diabres.2025.112454
- Sobhani NC. Impact of AID on Glycemic Profile and Maternal/Neonatal Outcomes in Pregnancy: A Review of the Evidence From Observational Studies. J Diabetes Sci Technol. Published online March 22, 2025:19322968251327603. doi:10.1177/19322968251327603
- Benhalima K, Durnwald C, Sweeting A, et al. Application of continuous glucose monitoring and automated insulin delivery technologies for pregnant women with type 1, type 2, or gestational diabetes: an international consensus statement. Lancet Diabetes Endocrinol. 2026;14(2):157-177. doi:10.1016/S2213-8587(25)00335-3

