We’re tackling common questions about automated insulin delivery and breaking down new consensus recommendations for use during pregnancy.
1. What is an AID system?
Automated insulin delivery (AID) systems consist of three main parts: a continuous glucose monitor (CGM), an insulin pump, and an algorithm. The CGM transmits real-time glucose data to an algorithm, which modulates how much insulin the pump delivers based on glucose trends and predictions.
2. How is AID different from other methods of insulin delivery?
For people on intensive insulin therapy, AID can dramatically reduce the amount of active effort and decision-making needed compared to other methods, such as MDI and open-loop insulin delivery.

3. Who is it for?
The 2026 American Diabetes Association Standards of Care recommend AID for:
- People with type 1 diabetes
- People with type 2 diabetes on basal + bolus insulin treatment
- People with other forms of insulin-deficient diabetes (LADA and others).
The ADA also recommends considering AID for people on basal insulin only, who are not meeting targets.1
Outside of pregnancy, all AID systems are approved for use in type 1 diabetes. For type 2 diabetes, Omnipod 5, MiniMed 780G, and t:Slim x2 or Mobi pumps with the Control-IQ+ algorithm are approved.
Research on AID in pregnancy has focused on people with type 1 diabetes. The 2026 consensus statement on AID and CGM use in pregnancy calls for much-needed research on AID in pregnancy with type 2 diabetes and does not make recommendations for AID use in this group due to the limited evidence. That said, all people with diabetes deserve an informed conversation about the tools available to them, regardless of their type of diabetes.
4. What are the benefits of AID?
Outside of pregnancy, studies show that AID systems are the best available tools to meet glucose targets, helping people with type 1 diabetes reach 12% higher time in range (70–180 mg/dL) on average than open-loop insulin pumps or multiple daily injections.2–4
In pregnancy with type 1 diabetes, randomized controlled trials have demonstrated that AID systems can increase time spent in the pregnancy target range (63–140 mg/dL) by up to 15% on average compared to MDI or open-loop pump therapy with a CGM.5,6 This is clinically relevant, as every additional 5% time in pregnancy range has been shown to reduce risk of adverse maternal and neonatal outcomes.7
5. Is AID safe to use during pregnancy?
Studies have found that some of the available AID systems can safely manage glucose during pregnancy with type 1 diabetes without increasing risk for dangerous lows or highs.5,8–13
As of 2026, three systems have been evaluated in large randomized controlled trials of pregnant patients with type 1 diabetes:
- MiniMed 780G (CRISTAL trial, using assistive techniques beyond standard use)9
- CamDiab CamAPS FX (AiDAPT trial)8
- Tandem t:Slim X2 with Control-IQ (CIRCUIT trial)5
This evidence provides greater confidence in the suitability of these devices for use in pregnancy. Because much of this research is new or ongoing, regulatory approvals are few and far between. Tandem Control IQ+ and CamAPS are the only AID algorithms FDA-approved for use in pregnancy, but CamAPS is not yet commercially available in the United States. Globally, several countries have approved CamAPS for use in pregnancy, and MiniMed 780G received a European conformity mark (CE) for use in type 1 diabetes pregnancy in Europe. No system has been approved specifically for pregnancy with type 2 diabetes. Still, many people with diabetes and their care teams have used the available systems during pregnancy and found them to be very helpful.
6. Which AID systems are recommended for pregnancy?
If possible, experts recommend using an AID system that meets the following three criteria:14
- Studied in a large randomized controlled trial of pregnant people
- Demonstrated >5% improvement in time in the target pregnancy range compared to non-AID methods of insulin delivery
- Has the ability to adapt to insulin sensitivity changes in pregnancy (either with a pregnancy-specific glucose target and/or algorithm features).
So, which systems meet these criteria?
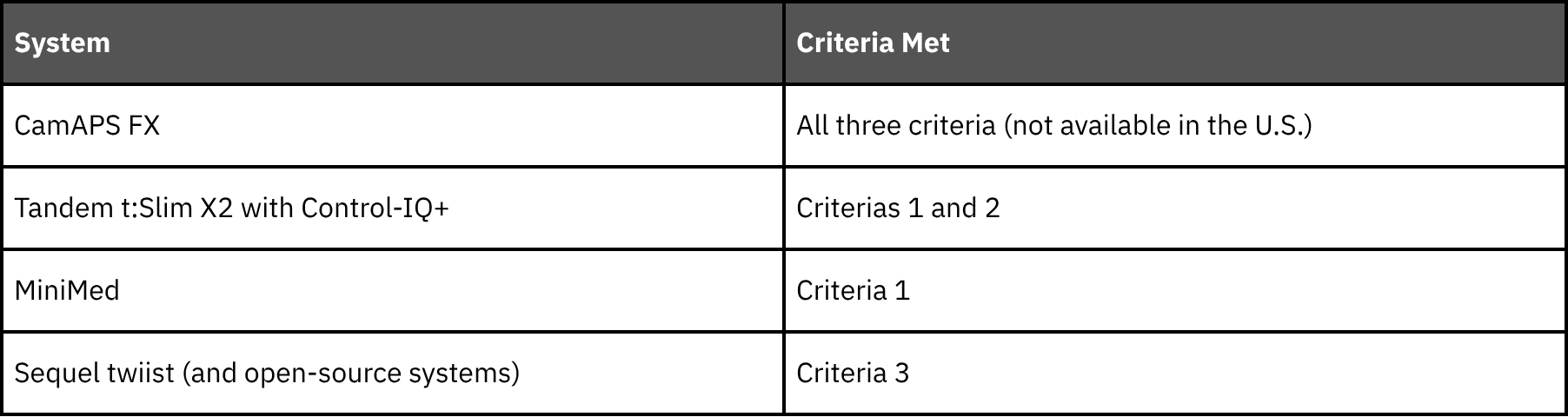
When discussing options, consider the degree to which settings and dosing strategies will need to be adjusted (beyond what is typically necessary outside of pregnancy), and whether the system has been shown to increase time in pregnancy range.
If someone in your care can’t access a system that meets all the recommended criteria, other AID systems may still be a practical choice for diabetes management during pregnancy. Setting adjustments and modified dosing strategies can optimize other systems for pregnancy needs, allowing individuals to stay on a device they’re comfortable with, or reap other benefits such as convenience. Providing (or referring to) individualized support to manage these changes is important. Some systems are more challenging to use since their lowest targets may be too high for pregnancy or they may have less adjustable settings. In these cases, consider switching to a different AID system or using the pump in manual mode if possible.
7. Can AID be initiated during pregnancy?
Yes! Experts recommend considering AID as early as possible in pregnancy, given the importance of careful management for maternal and neonatal health outcomes, and the proven benefits some systems provide. The sooner, the better, especially if not meeting pregnancy goals on current therapy.
References
- American Diabetes Association Professional Practice Committee for Diabetes*. 7. Diabetes Technology: Standards of Care in Diabetes—2026. Diabetes Care. 2025;49(Supplement_1):S150-S165. doi:10.2337/dc26-S007
- Boughton CK, Hovorka R. New closed-loop insulin systems. Diabetologia. 2021;64(5):1007-1015. doi:10.1007/s00125-021-05391-w
- Leelarathna L, Choudhary P, Wilmot EG, et al. Hybrid closed-loop therapy: Where are we in 2021? Diabetes Obes Metab. 2021;23(3):655-660. doi:10.1111/dom.14273
- Phillip M, Nimri R, Bergenstal RM, et al. Consensus Recommendations for the Use of Automated Insulin Delivery Technologies in Clinical Practice. Endocr Rev. 2023;44(2):254-280. doi:10.1210/endrev/bnac022
- Donovan LE, Lemieux P, Dunlop AD, et al. Closed-Loop Insulin Delivery in Type 1 Diabetes in Pregnancy: The CIRCUIT Randomized Clinical Trial. JAMA. Published online October 24, 2025. doi:10.1001/jama.2025.19578
- Lee TTM, Collett C, Bergford S, et al. Automated Insulin Delivery in Women with Pregnancy Complicated by Type 1 Diabetes. N Engl J Med. 2023;389(17):1566-1578. doi:10.1056/NEJMoa2303911
- Murphy HR. Continuous glucose monitoring targets in type 1 diabetes pregnancy: every 5% time in range matters. Diabetologia. 2019;62(7):1123-1128. doi:10.1007/s00125-019-4904-3
- Lee TT, Collett C, Bergford S, et al. Automated closed-loop insulin delivery for the management of type 1 diabetes during pregnancy: the AiDAPT RCT. Published online 2024. Accessed September 8, 2024. http://www.ncbi.nlm.nih.gov/books/NBK603527/
- Benhalima K, Beunen K, Van Wilder N, et al. Comparing advanced hybrid closed loop therapy and standard insulin therapy in pregnant women with type 1 diabetes (CRISTAL): a parallel-group, open-label, randomised controlled trial. Lancet Diabetes Endocrinol. 2024;12(6):390-403. doi:10.1016/S2213-8587(24)00089-5
- Tahir S, Naeem S, Nayyab I, et al. Hybrid closed loop insulin therapy versus standard therapy in pregnant women with type 1 diabetes: A systematic review and meta-analysis of randomized controlled trials. Eur J Obstet Gynecol Reprod Biol. 2025;310:113969. doi:10.1016/j.ejogrb.2025.113969
- Stamati A, Christoforidis A. Automated insulin delivery in pregnant women with type 1 diabetes mellitus: a systematic review and meta-analysis. Acta Diabetol. 2025;62(4):441-452. doi:10.1007/s00592-025-02446-x
- Pham AQ, Lingvay I, Ahmadi N, Santiago-Munoz PC, Abreu M. Use of commercially available automated insulin delivery systems in pregnant people with type 1 diabetes. Diabetes Res Clin Pract. 2025;228:112454. doi:10.1016/j.diabres.2025.112454
- Sobhani NC. Impact of AID on Glycemic Profile and Maternal/Neonatal Outcomes in Pregnancy: A Review of the Evidence From Observational Studies. J Diabetes Sci Technol. Published online March 22, 2025:19322968251327603. doi:10.1177/19322968251327603
- Benhalima K, Durnwald C, Sweeting A, et al. Application of continuous glucose monitoring and automated insulin delivery technologies for pregnant women with type 1, type 2, or gestational diabetes: an international consensus statement. Lancet Diabetes Endocrinol. 2026;14(2):157-177. doi:10.1016/S2213-8587(25)00335-3

